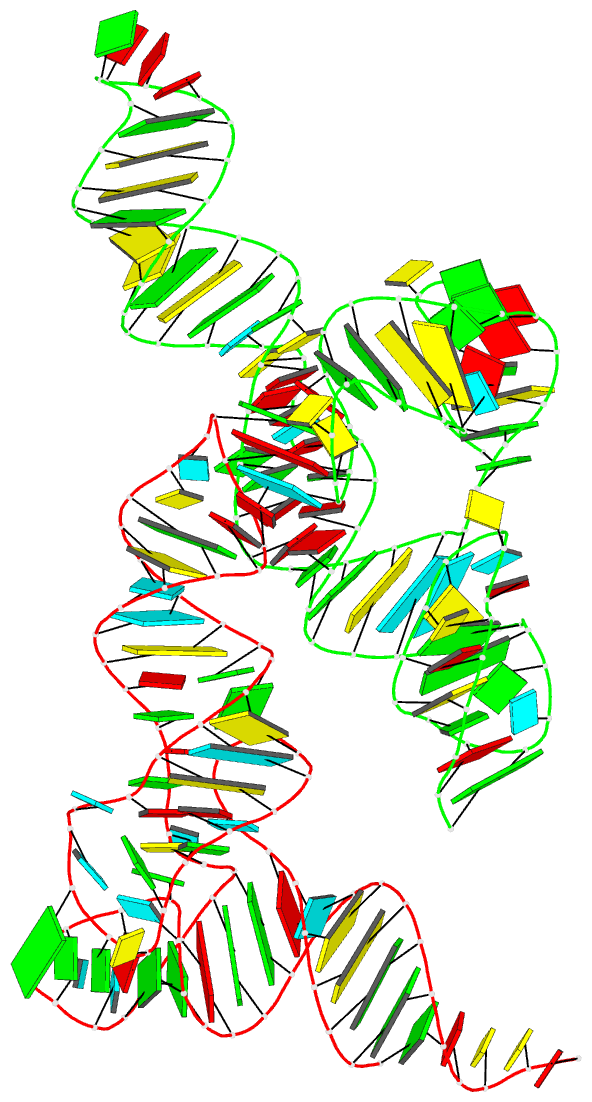
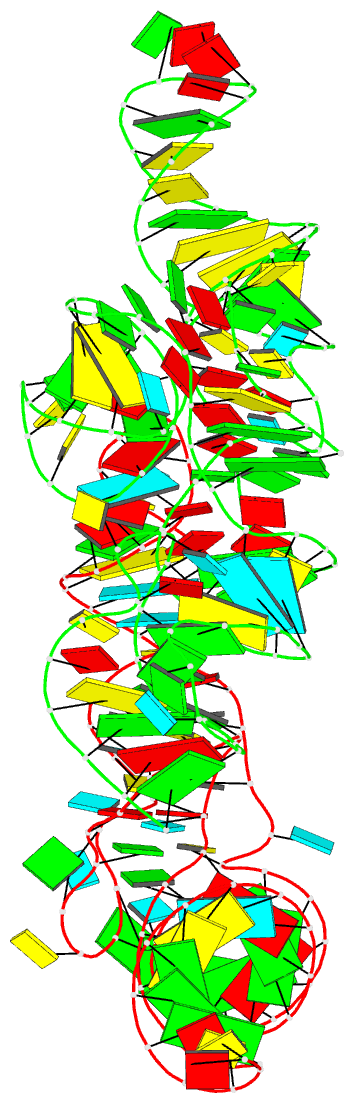
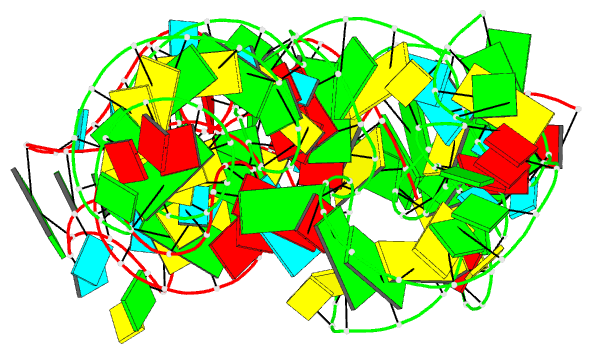
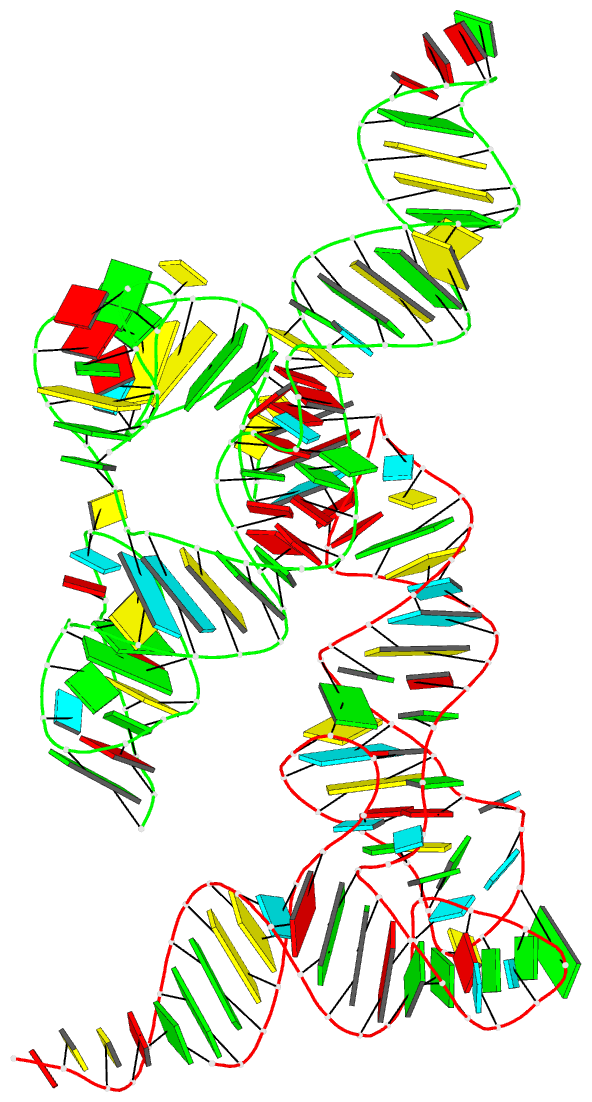
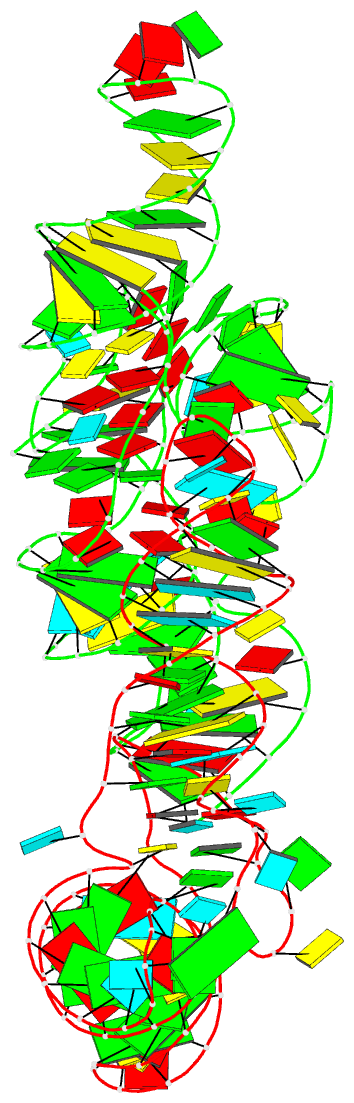
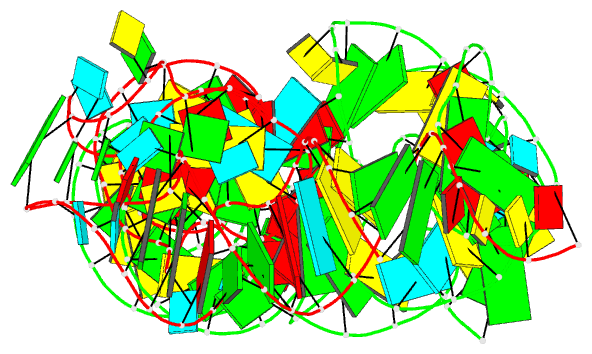
Summary information and primary citation
- PDB-id
- 6ufm; DSSR-derived features in text and JSON formats
- Class
- RNA
- Method
- X-ray (2.82 Å)
- Summary
- Cocrystal structure of the nocardia farcinica iles t-box riboswitch in complex with its cognate trna
- Reference
- Suddala KC, Zhang J (2019): "High-affinity recognition of specific tRNAs by an mRNA anticodon-binding groove." Nat.Struct.Mol.Biol., 26, 1114-1122. doi: 10.1038/s41594-019-0335-6.
- Abstract
- T-box riboswitches are modular bacterial noncoding RNAs that sense and regulate amino acid availability through direct interactions with tRNAs. Between the 5' anticodon-binding stem I domain and the 3' amino acid sensing domains of most T-boxes lies the stem II domain of unknown structure and function. Here, we report a 2.8-Å cocrystal structure of the Nocardia farcinica ileS T-box in complex with its cognate tRNAIle. The structure reveals a perpendicularly arranged ultrashort stem I containing a K-turn and an elongated stem II bearing an S-turn. Both stems rest against a compact pseudoknot, dock via an extended ribose zipper and jointly create a binding groove specific to the anticodon of its cognate tRNA. Contrary to proposed distal contacts to the tRNA elbow region, stem II locally reinforces the codon-anticodon interactions between stem I and tRNA, achieving low-nanomolar affinity. This study illustrates how mRNA junctions can create specific binding sites for interacting RNAs of prescribed sequence and structure.