Summary information and primary citation
- PDB-id
- 6a5r; DSSR-derived features in text and JSON formats
- Class
- transcription-RNA-DNA
- Method
- cryo-EM (8.7 Å)
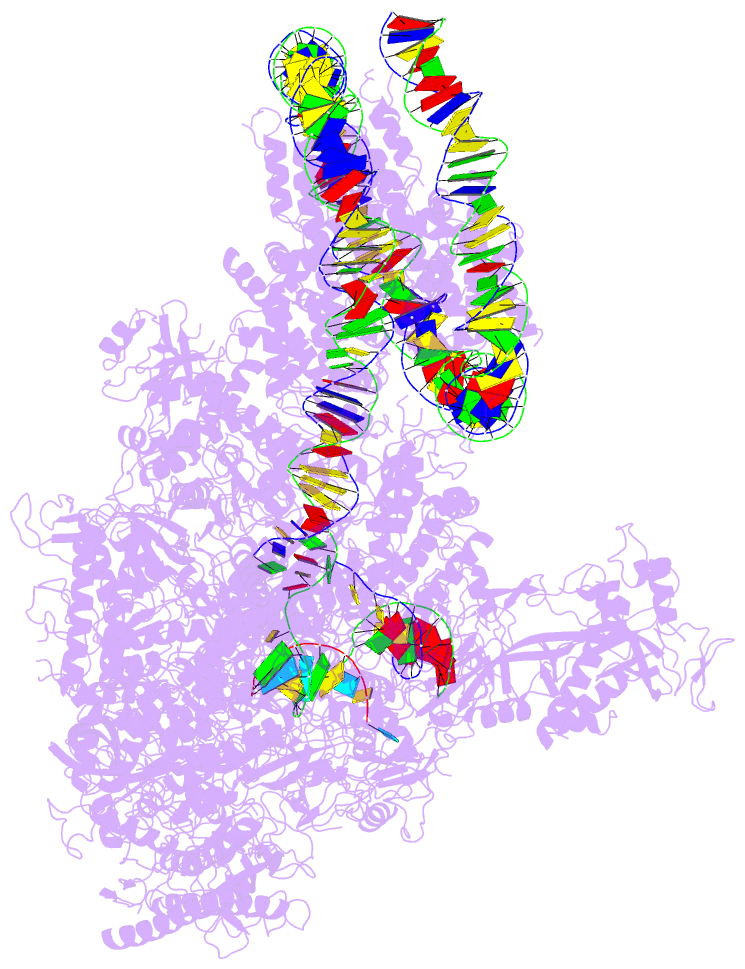
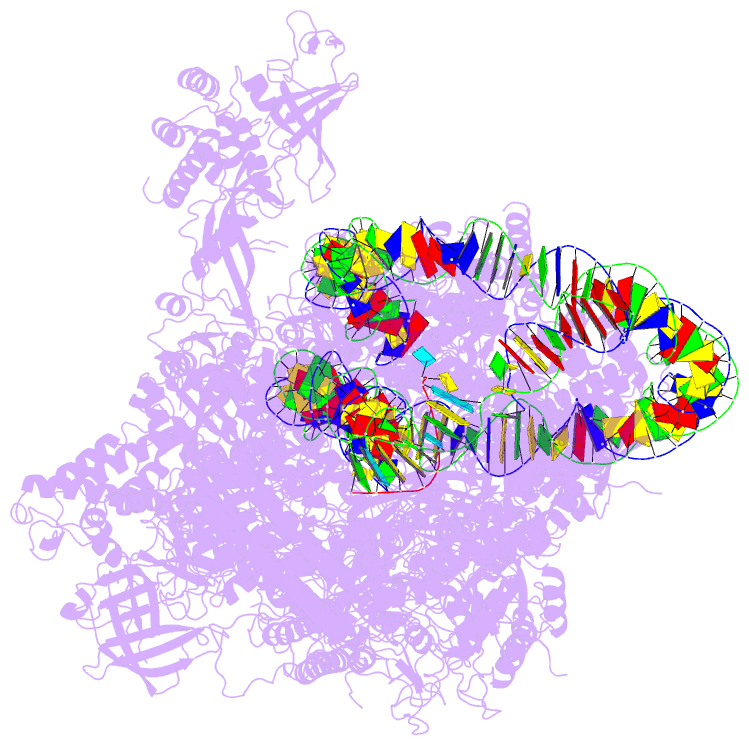
- Summary
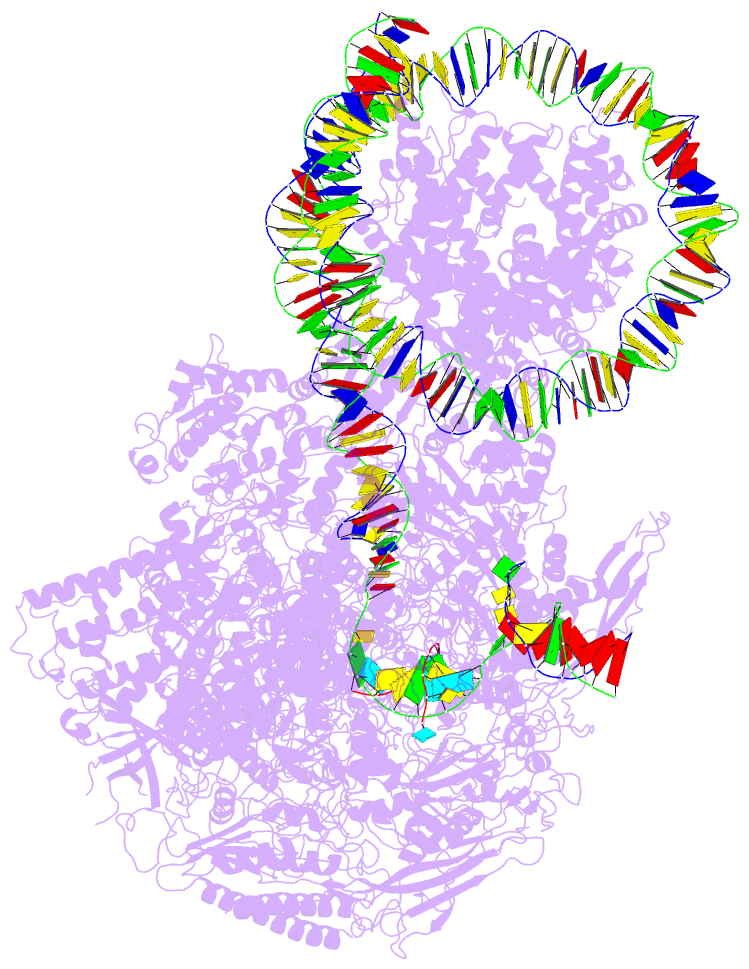
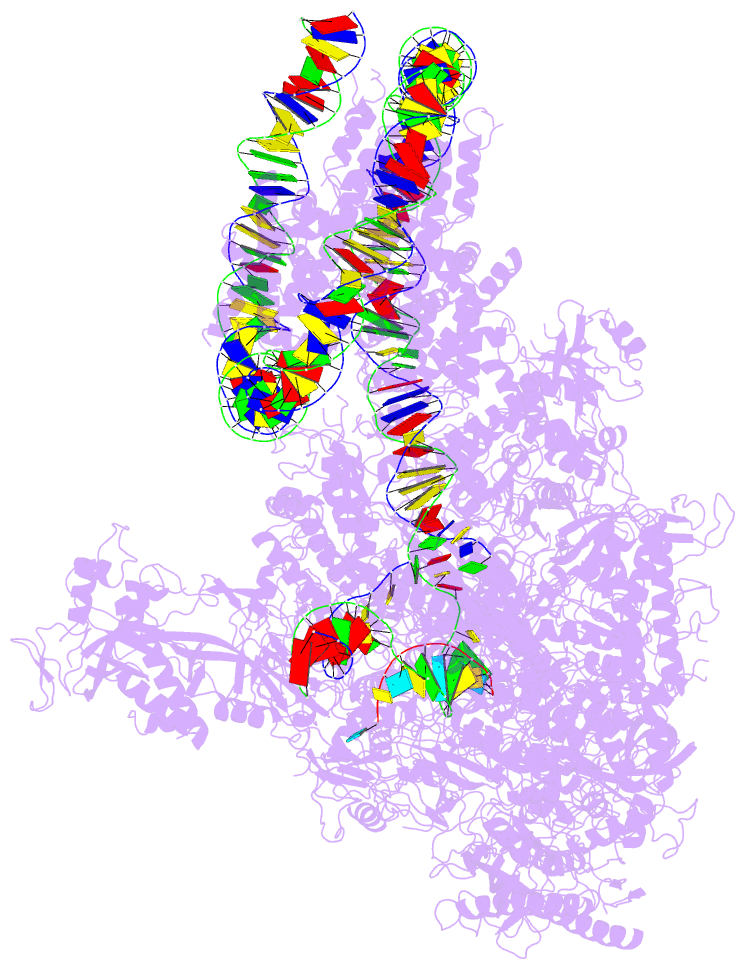
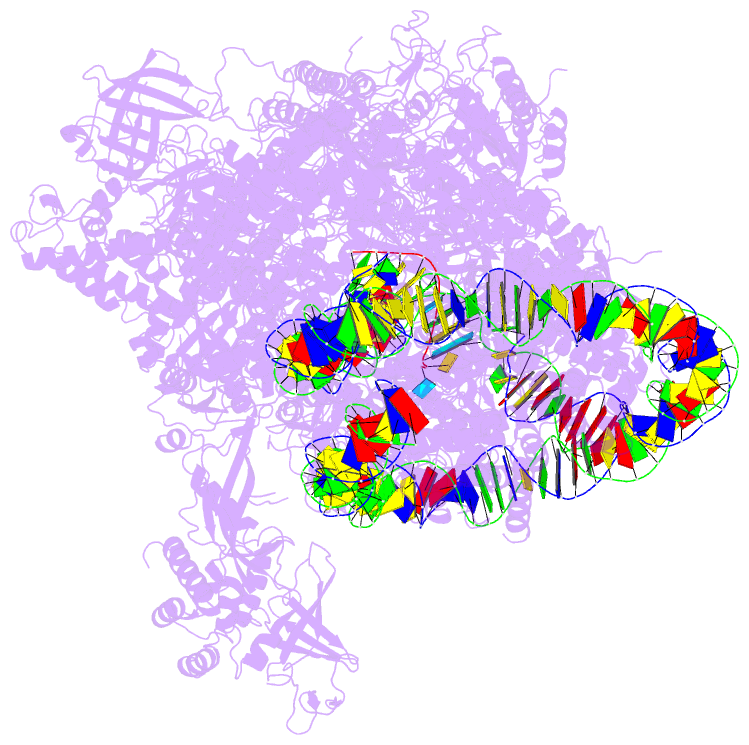
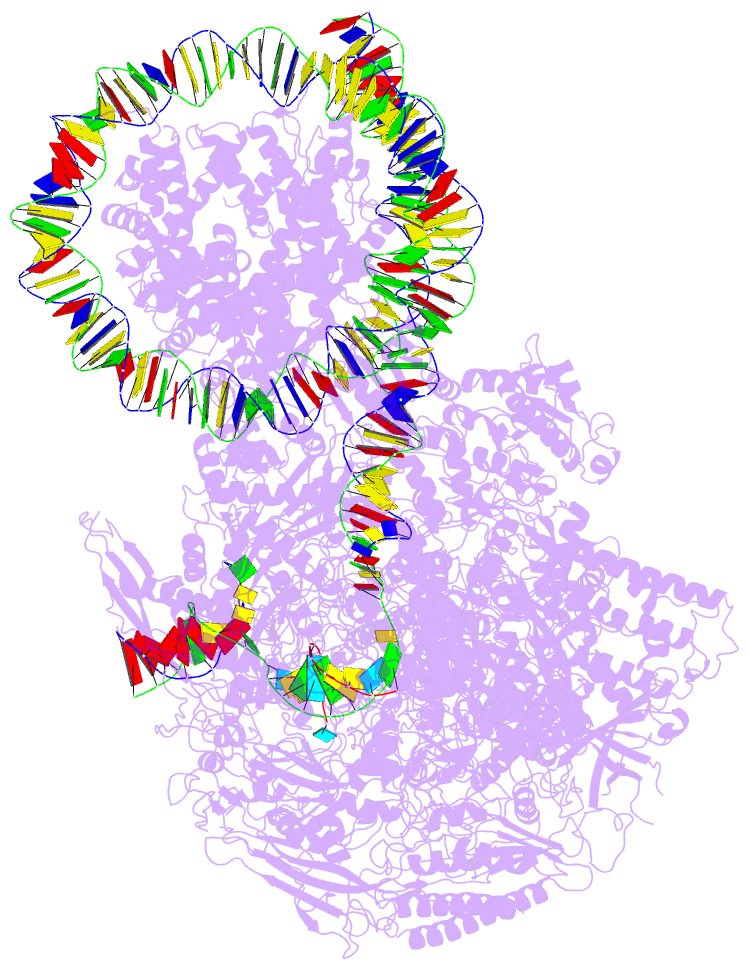
- RNA polymerase ii elongation complex stalled at shl(-2) of the nucleosome
- Reference
- Kujirai T, Ehara H, Fujino Y, Shirouzu M, Sekine SI, Kurumizaka H (2018): "Structural basis of the nucleosome transition during RNA polymerase II passage." Science, 362, 595-598. doi: 10.1126/science.aau9904.
- Abstract
- Genomic DNA forms chromatin, in which the nucleosome is the repeating unit. The mechanism by which RNA polymerase II (RNAPII) transcribes the nucleosomal DNA remains unclear. Here we report the cryo-electron microscopy structures of RNAPII-nucleosome complexes, in which RNAPII pauses at the superhelical locations, SHL(-6), SHL(-5), SHL(-2), and SHL(-1), of the nucleosome. RNAPII pauses at the major histone-DNA contact sites, and the nucleosome interactions with the RNAPII subunits stabilize the pause. These structures reveal snapshots of nucleosomal transcription, where RNAPII gradually tears DNA from the histone surface, while preserving the histone octamer. Interestingly, the nucleosomes in the SHL(-1) complexes are bound to a "foreign" DNA segment, which might explain the histone transfer mechanism. These results provide the foundations for understanding chromatin transcription and epigenetic regulation.