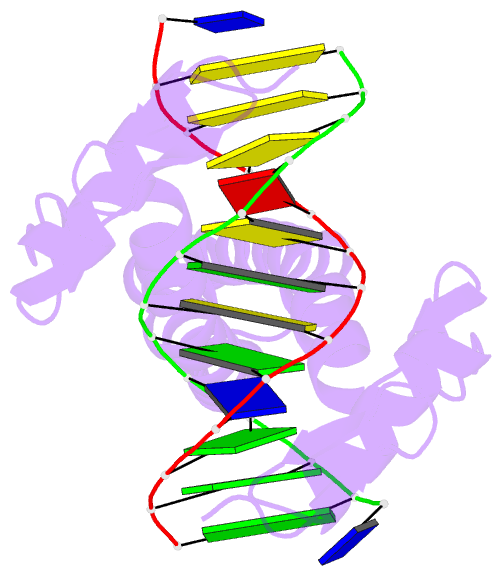
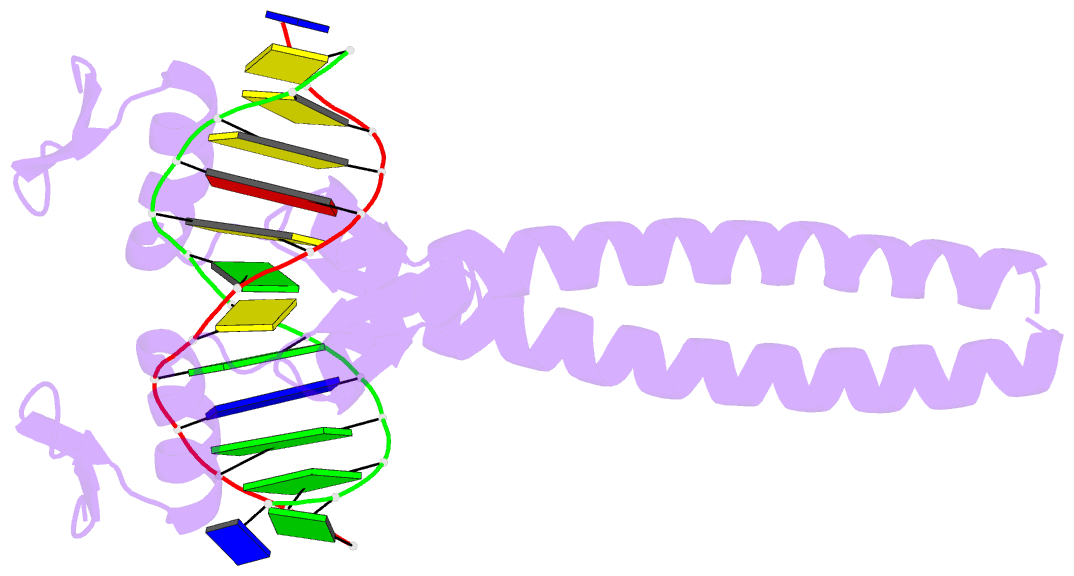
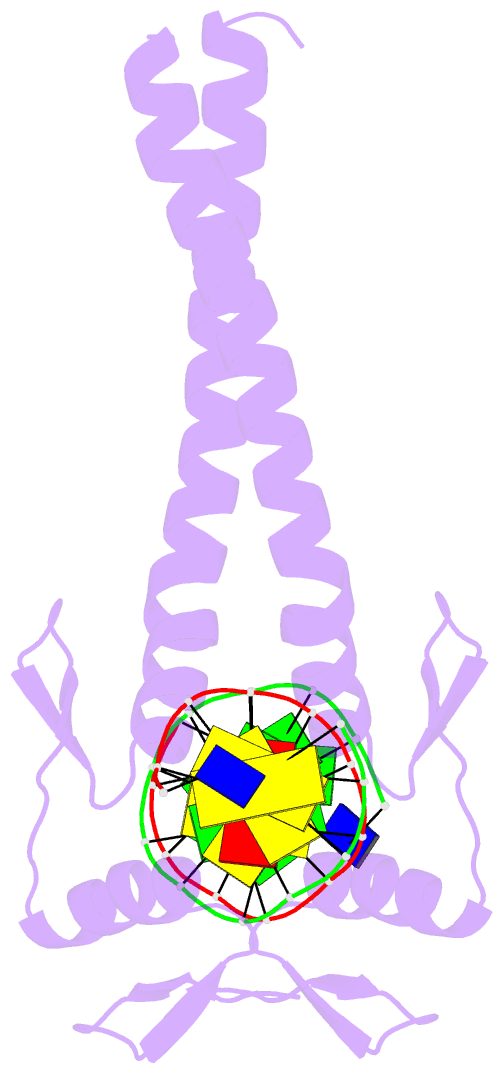
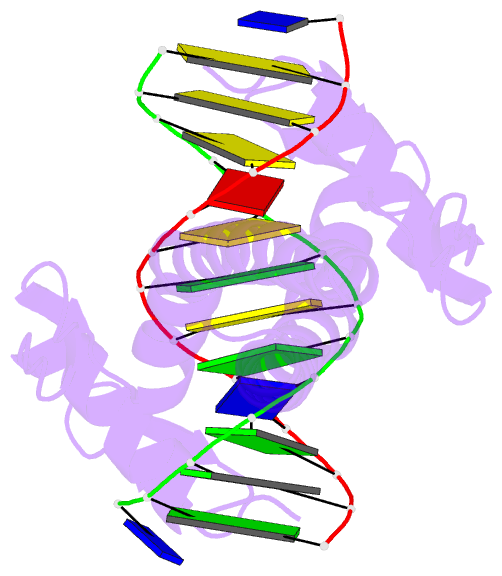
Summary information and primary citation
- PDB-id
- 1llm; DSSR-derived features in text and JSON formats
- Class
- transcription-DNA
- Method
- X-ray (1.5 Å)
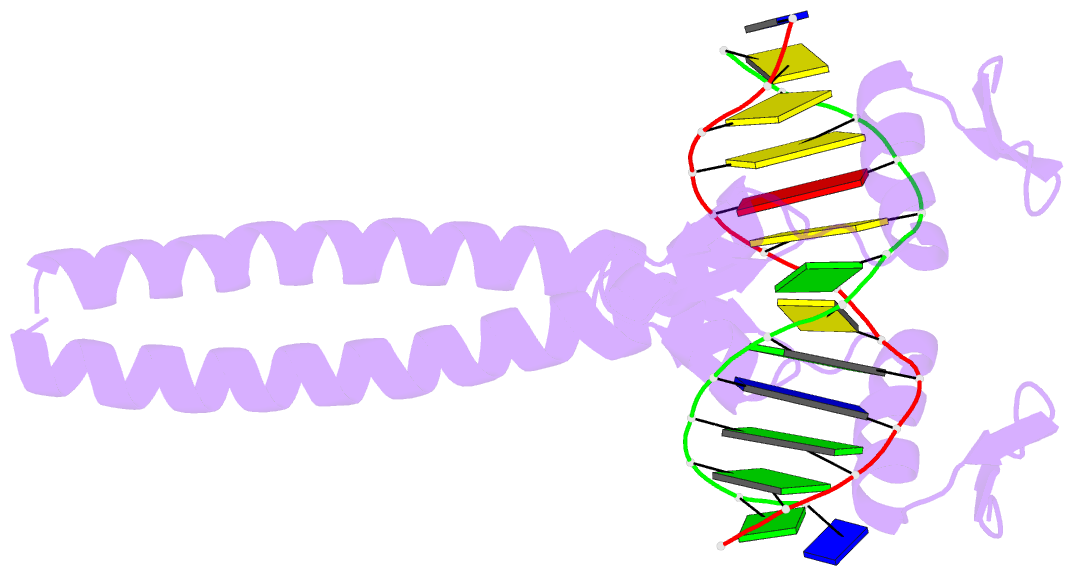
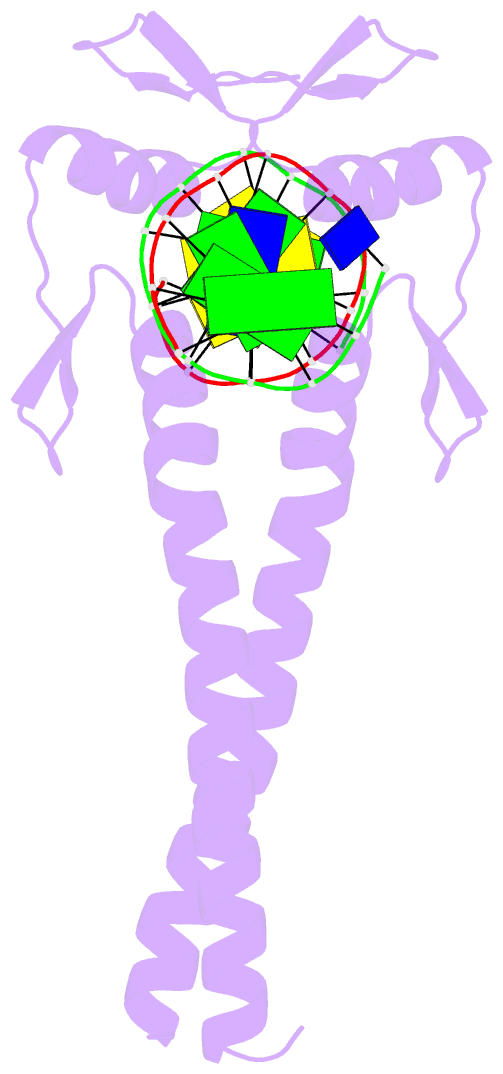
- Summary
- Crystal structure of a zif23-gcn4 chimera bound to DNA
- Reference
- Wolfe SA, Grant RA, Pabo CO (2003): "Structure of a designed dimeric zinc finger protein bound to DNA." Biochemistry, 42, 13401-13409. doi: 10.1021/bi034830b.
- Abstract
- Proteins that employ dimerization domains to bind cooperatively to DNA have a number of potential advantages over monomers with regards to gene regulation. Using a combination of structure-based design and phage display, a dimeric Cys(2)His(2) zinc finger protein has been created that binds cooperatively to DNA via an attached leucine zipper dimerization domain. This chimera, derived from components of Zif268 and GCN4, displayed excellent DNA-binding specificity, and we now report the 1.5 A resolution cocrystal structure of the Zif268-GCN4 homodimer bound to DNA. This structure shows how phage display has annealed the DNA binding and dimerization domains into a single functional unit. Moreover, this chimera provides a potential platform for the creation heterodimeric zinc finger proteins that can regulate a desired target gene through cooperative DNA recognition.