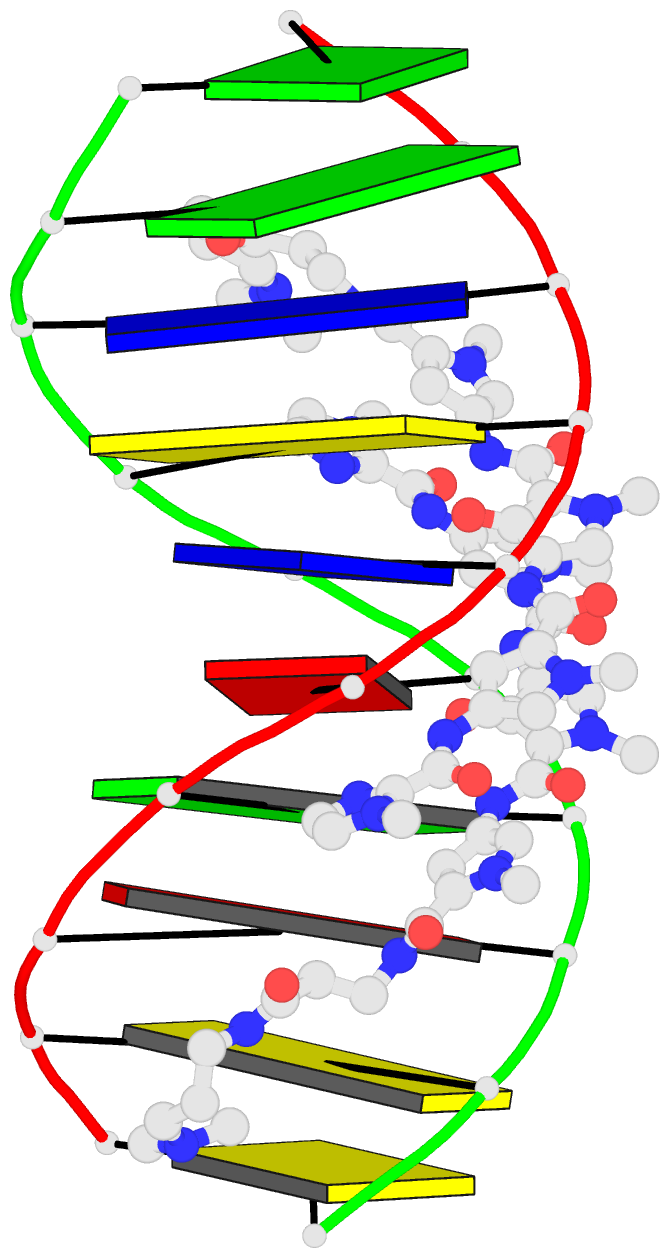
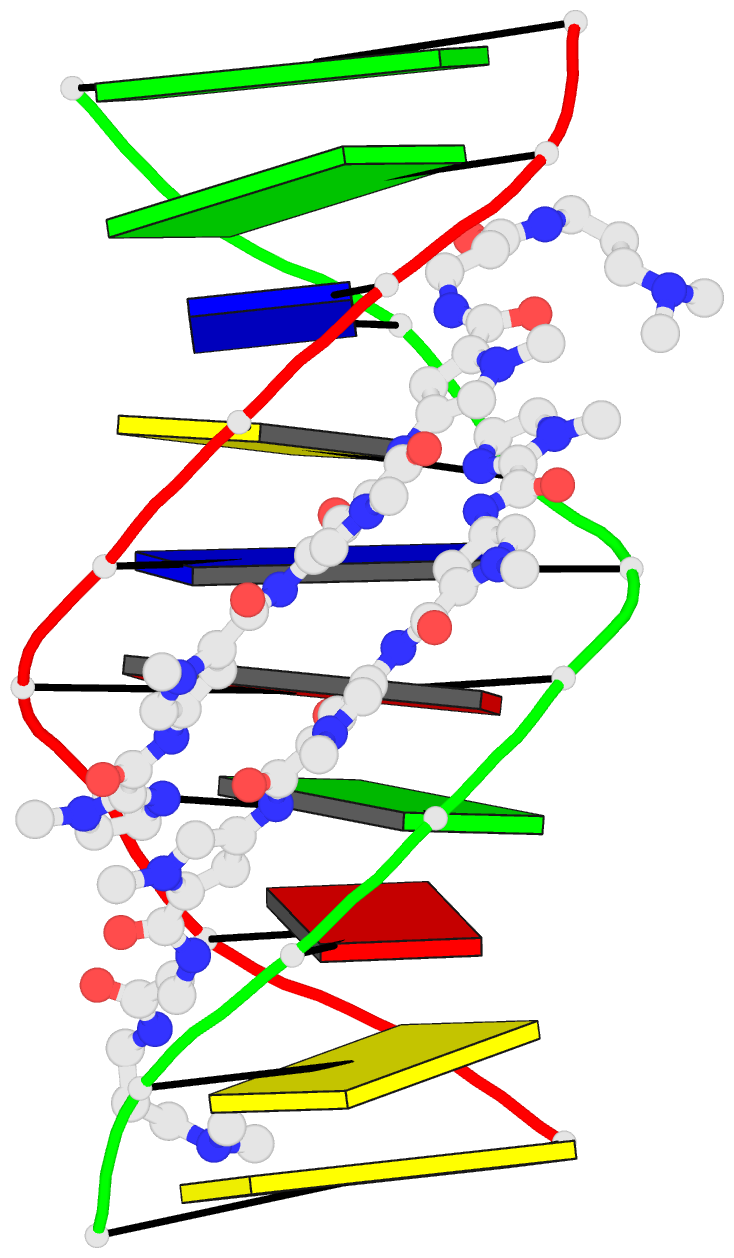
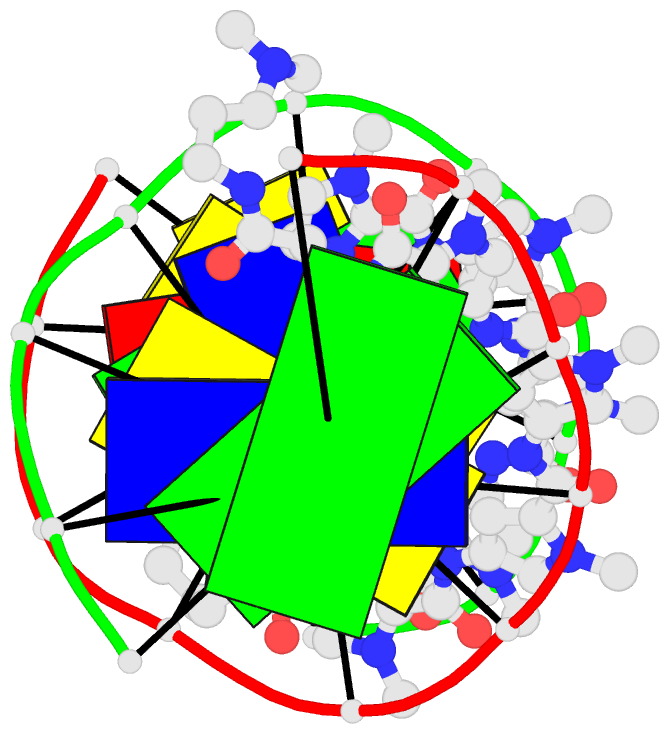
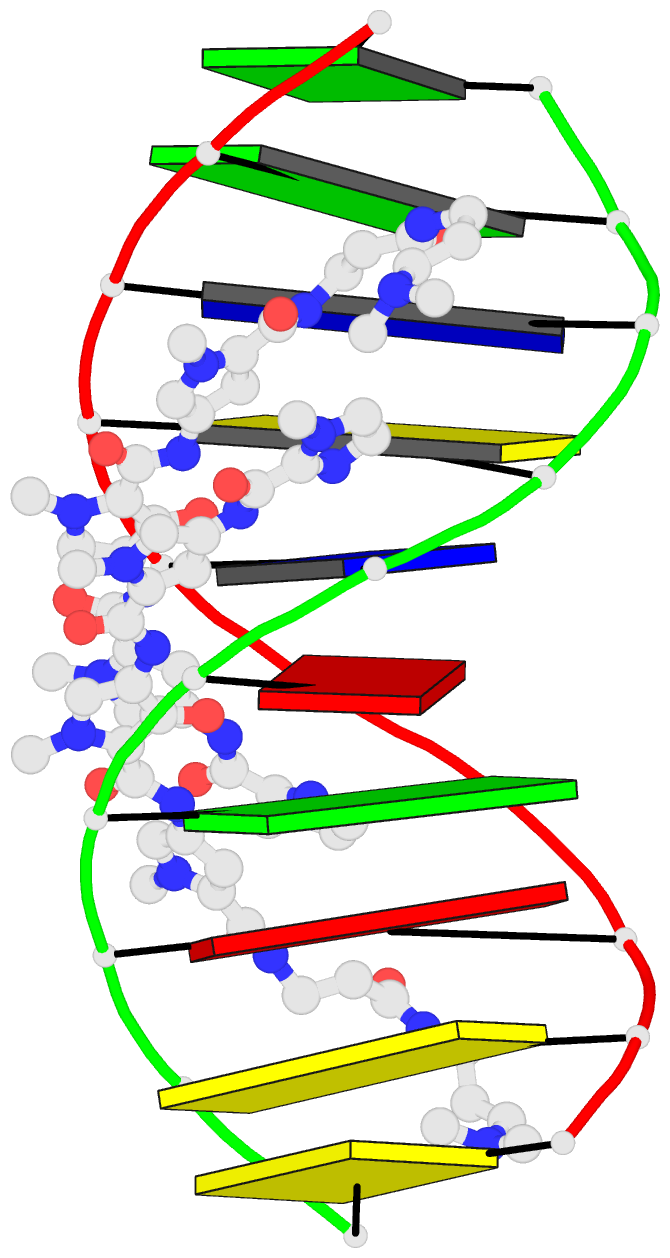
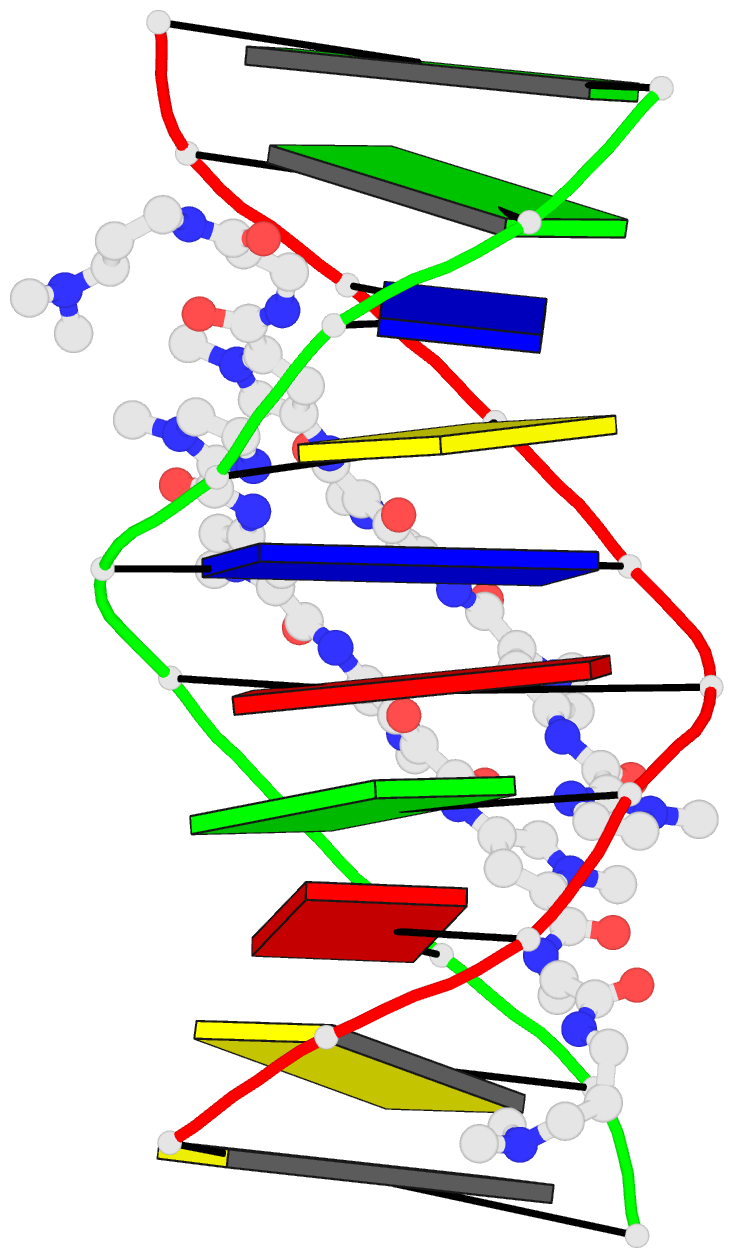
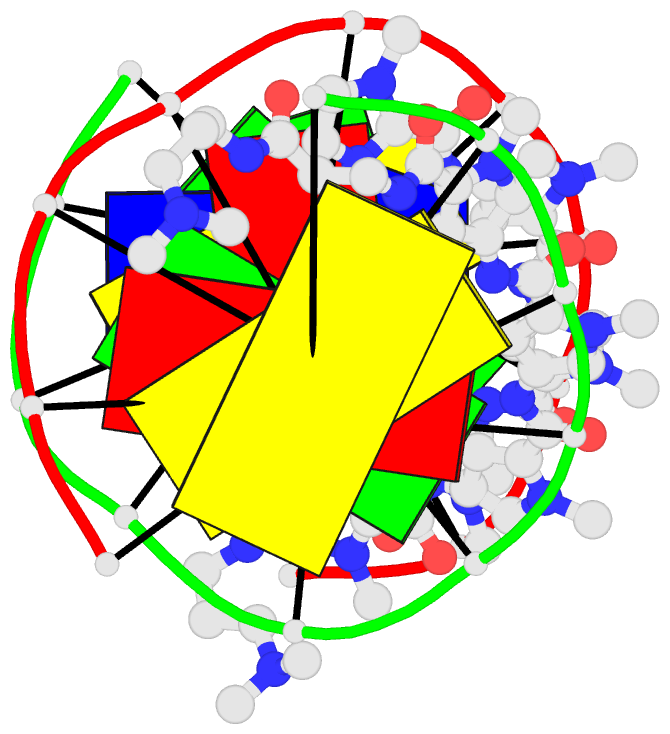
Summary information and primary citation
- PDB-id
- 1cvx; DSSR-derived features in text and JSON formats
- Class
- DNA
- Method
- X-ray (2.27 Å)
- Summary
- Crystal structure of polyamide dimer (impyhppybetadp)2 bound to b-DNA decamer ccagatctgg
- Reference
- Kielkopf CL, Bremer RE, White S, Szewczyk JW, Turner JM, Baird EE, Dervan PB, Rees DC (2000): "Structural effects of DNA sequence on T.A recognition by hydroxypyrrole/pyrrole pairs in the minor groove." J.Mol.Biol., 295, 557-567. doi: 10.1006/jmbi.1999.3364.
- Abstract
- Synthetic polyamides composed of three types of aromatic amino acids, N-methylimidazole (Im), N-methylpyrrole (Py) and N-methyl-3-hydroxypyrrole (Hp) bind specific DNA sequences as antiparallel dimers in the minor groove. The side-by-side pairings of aromatic rings in the dimer afford a general recognition code that allows all four base-pairs to be distinguished. To examine the structural consequences of changing the DNA sequence context on T.A recognition by Hp/Py pairs in the minor groove, crystal structures of polyamide dimers (ImPyHpPy)(2) and the pyrrole counterpart (ImPyPyPy)(2) bound to the six base-pair target site 5'-AGATCT-3' in a ten base-pair oligonucleotide have been determined to a resolution of 2.27 and 2.15 A, respectively. The structures demonstrate that the principles of Hp/Py recognition of T.A are consistent between different sequence contexts. However, a general structural explanation for the non-additive reduction in binding affinity due to introduction of the hydroxyl group is less clear. Comparison with other polyamide-DNA cocrystal structures reveals structural themes and differences that may relate to sequence preference.